- Blog
- Solving quadratic equations quiz
- Happy planner expense tracker half sheet printable
- 3d comic the chaperone- episode 5 free
- Siberian mouse girls xxx
- Online sketchbook free
- Super smash bros brawl wii download wbfs
- Commercial alarm monitoring invoice template
- Match baby animals to adults game
- Periodic table color code
- Bluestacks emulator pc download
- Avery 18163 word label template
- Household budget template percentages
- Scattergories questions lists downloadable free pintrest
- 2048 2048 game online free
- Directx 11 windows 10 download 64 bit
- Best valorant crosshair reddit
- Sketchup pro 2018 vray crack
- Gang gang cockatoo pet
- The game ratty catty
- Weight tracker template
- Periodic table molar mass in molesliters
- Stanley electric sharpshooter staple gun instructions
- Avengers infinity war myrtle beach sc
- Terraria free download pc games download
- Best free resume templates 2020
- Image converter to 128x128
- Track ip address of a facebook profile
- Linkedin logo color png transparent
- Skyrim sexlab aroused redux for vanilla skyrim
- Convert webp gif
- 3d invigorator photoshop plugin torrent mac
- Create a signature in pdf
- Tri color hair highlights ideas asian men
- Water density gml
- Hearts card game free online
- Initial d psp english iso
- Total war rome 2 faction
- Farming simulator 19 enter other players cars
- Challenge code omnisphere 2
- Is decibels a logarithmic scale
- Free resume template for registered nurse
- Twilight forest mod download for minecraft pe
(1)įor example, at 25☌ the density of metallic lead is 11.4 g mL -1 while the density of non-metallic sulfur is only 2.0 g mL -1. In general, metals are more dense than non-metals. This property of carbon dioxide gas has been used to fight fires, firstly because carbon dioxide gas doesn't burn, and secondly because the carbon dioxide gas can form a floating barrier that blocks off the supply of oxygen gas so that the substance can no longer burn. If you filled a balloon with pure carbon dioxide gas it will not float up, it will sink down to the floor! The density of carbon dioxide gas is about 0.0018 g mL -1.Ĭarbon dioxide gas is more dense than the air around you. If you fill a balloon with helium gas you need to hold onto the string otherwise it will float up and away! Helium gas is less dense than the air around you. The density of helium gas is about 0.00016 g mL -1. The density of the air you breath is about 0.001 g mL -1. The density of gases can also be measured. You may have seen pictures of oil spills from ships at sea in which you can see the black oil floating on top of the water, and then washing up onto the shore.īecause the oil floats on top of water, it is possible to contain the oil spill at sea before it washes ashore by building a floating wall around it then "scooping up" the oil from the surface. They are both less dense than water so they will also float on top of water. Oil and petrol (gasoline) do not mix with water. This concept of separating out layers of liquids based on their density is very useful in chemistry. If you are very careful, you can "pour off", or " decant", the vegetable oil leaving the water in the beaker. You will find the layer of more dense water at the bottom of the beaker and the less dense vegetable oil layer lying above the water. So, if you pour water and vegetable oil into the same beaker they will not mix but they will separate out into 2 layers. Liquid water is more dense than vegetable oil. If you weigh 1.00 mL of vegetable oil it might have a mass of about 0.8 g so its density is about 0.8 g/mL or 0.8 g ml -1. If you weigh 1.00 mL of liquid water it will have a mass of about 1 g so its density is about 1 g/mL or 1 g mL -1. You could use a 1.00 mL pipette to acquire a sample of the liquid and weigh this. If you want to measure the density of liquids in the lab, it is much easier to use units of mass per unit volume in millilitres.

You can use these cubes to determine the densities of some different solid substances like copper, iron, zinc etc. You probably have some 1 cm 3 cubes of different substances in your lab. Gold has a density of 19.3 grams per cubic centimeter, that is, 19.3 g/cm 3 or 19.3 g cm -3.Īluminium has a density of 2.7 grams per cubic centimeter, that is, 2.7 g/cm 3 or 2.7 g cm -3. The density of each substance is given in units of mass per unit volume, for our cubes this will be mass in grams per cubic centimeter, that is, g/cm -3 or g cm -3. We can also say that aluminium is less dense than gold because, for the same volume of the two substances, this volume of aluminium has less mass. Gold is said to be more dense than aluminium because, when we compare the same volume of the two different substances, this volume of gold has the greater mass. The other cube is made out of aluminium has a mass of only 2.7 grams. One cube is made out of gold and has a mass of 19.3 grams. Each cube measures 1 cm × 1 cm × 1cm so that each cube has a volume of 1 cm 3.

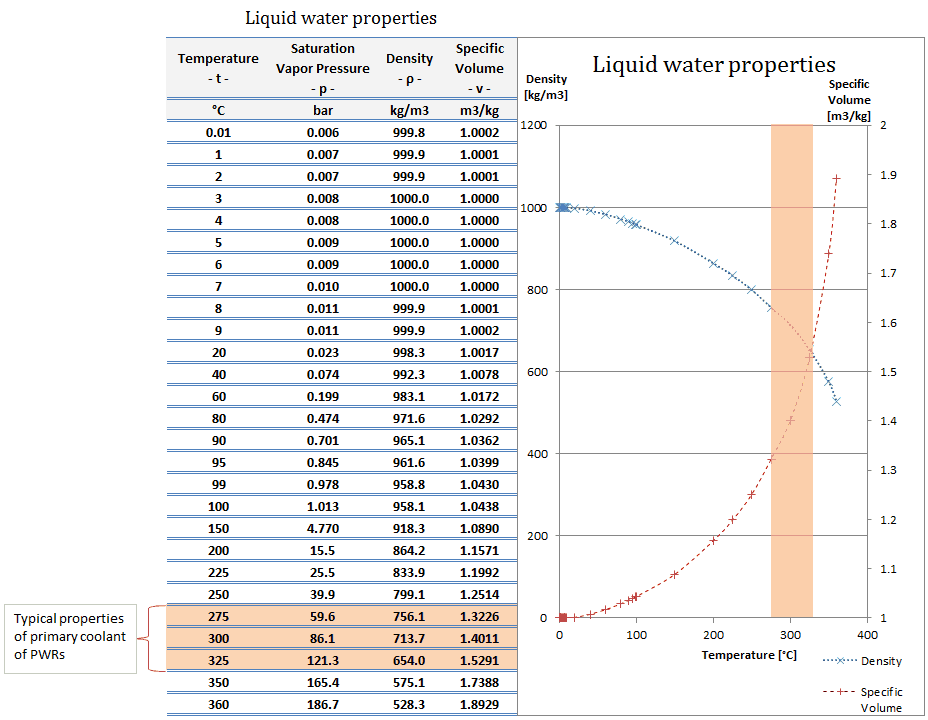
The metallic elements aluminium and gold are both solids at room temperature and pressure.īy weighing a known volume of metal we can determine the density of each metal. You can determine, or measure, the density of solids, liquids and gases in the lab. Journal of Physical and Chemical Reference Data 31, 387, 2002 DOI 10.1063/1.1461829.Density is defined as the amount of mass per unit volume. Pruß: The IAPWS Formulation 1995 for the Thermodynamic Properties of Ordinary Water Substance for General and Scientific Use. * - NIST Standard Reference Database 69: NIST Chemistry WebBook, Thermophysical Properties of Fluid Systems, Isobaric Properties for Water, 2008. * Temperature according to ITS-90 (International Temperature Scale from 1990). The following table lists the density D of water as a function of the temperature under isobaric conditions : The density of water (H 2O) changes depending on temperature and pressure - as with other substances.
- Blog
- Solving quadratic equations quiz
- Happy planner expense tracker half sheet printable
- 3d comic the chaperone- episode 5 free
- Siberian mouse girls xxx
- Online sketchbook free
- Super smash bros brawl wii download wbfs
- Commercial alarm monitoring invoice template
- Match baby animals to adults game
- Periodic table color code
- Bluestacks emulator pc download
- Avery 18163 word label template
- Household budget template percentages
- Scattergories questions lists downloadable free pintrest
- 2048 2048 game online free
- Directx 11 windows 10 download 64 bit
- Best valorant crosshair reddit
- Sketchup pro 2018 vray crack
- Gang gang cockatoo pet
- The game ratty catty
- Weight tracker template
- Periodic table molar mass in molesliters
- Stanley electric sharpshooter staple gun instructions
- Avengers infinity war myrtle beach sc
- Terraria free download pc games download
- Best free resume templates 2020
- Image converter to 128x128
- Track ip address of a facebook profile
- Linkedin logo color png transparent
- Skyrim sexlab aroused redux for vanilla skyrim
- Convert webp gif
- 3d invigorator photoshop plugin torrent mac
- Create a signature in pdf
- Tri color hair highlights ideas asian men
- Water density gml
- Hearts card game free online
- Initial d psp english iso
- Total war rome 2 faction
- Farming simulator 19 enter other players cars
- Challenge code omnisphere 2
- Is decibels a logarithmic scale
- Free resume template for registered nurse
- Twilight forest mod download for minecraft pe
